The goal of my current research is to understand how motor circuits generating body movements develop and mature, and apply the knowledge to protect them from aging and disease. To achieve this goal, we primarily study the motor systems of zebrafish because they allow access to motor circuits at the behavioral, cellular and genetic/molecular levels in the living animal.
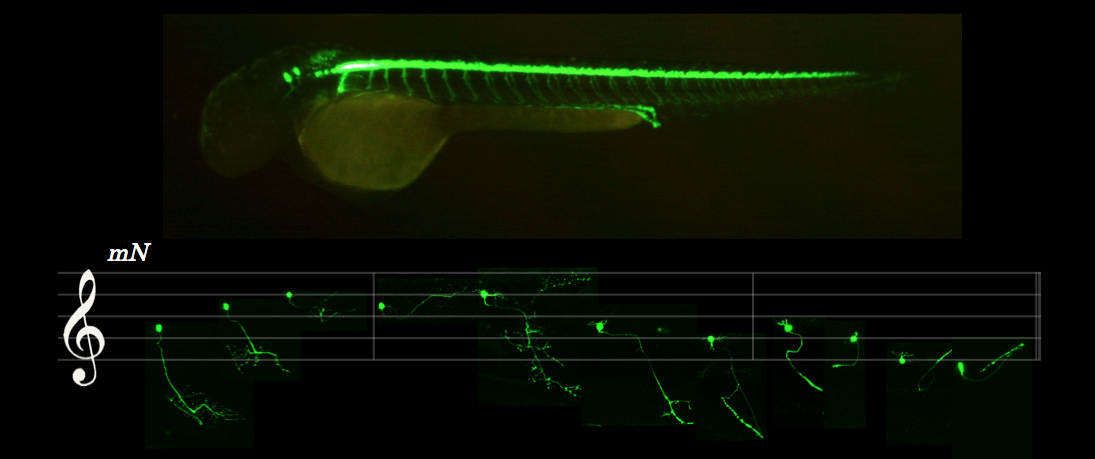
Disease of motor system

Amyotrophic lateral sclerosis (ALS) is a neurological disorder in which the upper and lower motor neurons progressively degenerate, leading to muscular atrophy and eventually fatal paralysis. We address when and how healthy motor neurons begin to become abnormal and pathological in ALS by combining genetics, in vivo cell biology and systems biology of the crystal clear zebrafish neuromuscular system. Primary focus is on RNA-binding proteins that maintain physiological homeostasis of motor neurons. (See Asakawa et al., Nat Commun 2020) Key words: TDP-43, Optogenetics, Phase separation
Development of motor system
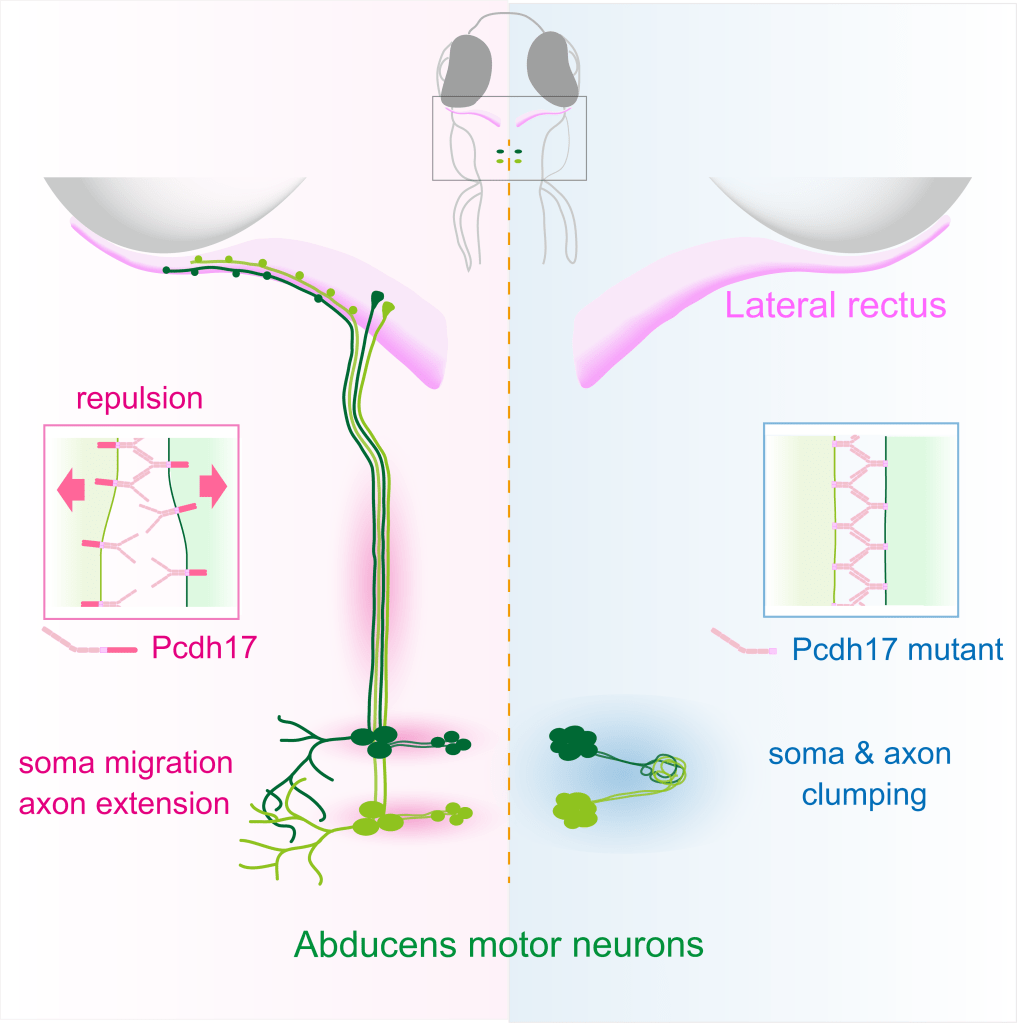
Diverse movements of our body are regulated by motor neurons aligned in the brainstem and spinal cord that connect with cranial and skeletal muscles, respectively. Developing motor neurons with similar functions cluster together into discrete nuclei or columns, and collectively projected onto peripheral target muscles often in a topographic manner. We address cellular and molecular mechanisms underlying motor nucleus/column organizations and their faithful projections to target muscles. (See Asakawa and Kawakami, Cell Reports 2018) Key words: Cell size, Protocadherin, Brazil nut effect
Dissection of motor system

A comprehensive understanding of how the nervous system generates behavior depends in large part on knowing how the system is organized. Among the major challenges which remain is the identification of the functional neural circuits that produce measureable behavioral outputs from the diverse anatomical data and also the genetic programs that assemble these circuits. To achieve this end, we develop methods for visualizing and manipulating functional motor circuits as well as analyzing the gene functions that allow motor circuits to operate. (See Asakawa et al., PNAS 2008, Asakawa et al., Front Neural Circuits 2013) Key words: Transgenic, Gal4/UAS, BAC transgegesis
Selected publications (→ Full publication list)
Multi-phaseted problems of TDP-43 in selective neuronal vulnerability in ALS
Asakawa K*, Handa H, Kawakami K.
Cellular and Molecular Life Sciences (2021). https://doi.org/10.1007/s00018-021-03792-z (*Correspondence)
Asakawa K*, Handa H, Kawakami K*.
Nature Communications 11:1004 (2020) (*Co-correspondence)
Asakawa K* and Kawakami K*.
Cell Reports 24:1562-1572 (2018) (*Co-correspondence)
Asakawa K*, Gembu A and Kawakami K*.
Frontiers in Neural Circuits 7:100 (2013) (*Co-correspondence)
An mnr2b/hlxb9lb enhancer trap line that labels spinal and abducens motor neurons in zebrafish.
Asakawa K, Higashijima S, and Kawakami K.
Developmental Dynamics 241, 327-332 (2012)
Asakawa K, Suster ML, Mizusawa K, Nagayoshi S, Kotani T, Urasaki A, Kishimoto Y, Hibi M and Kawakami K.
Proc Natl Acad Sci U S A 105:1255-1260 (2008)
Funding
公的研究費
2023-24 科研費 学術変革(A) 公募 (代表)
2022-23 科研費・挑戦的研究(萌芽) (代表)
2022-23 科研費 新学術領域研究 公募 (代表)
2021-25 AMED-PRIME(代表)
2022-25 科研費 基盤研究(B)(代表)
2020-21 科研費 新学術領域研究 公募 (代表)
2019-21 科研費 基盤研究(C)(代表)
2016-18 科研費 基盤研究(C)(代表)
2013-15 科研費 若手研究(B)(代表)
2011-12 科研費 新学術領域研究 公募 (代表)
2010-12 科研費 若手研究(B)(代表)
2010-12 科研費 若手研究(B)(代表)
助成金
2023 中外創薬科学財団・研究助成
2022 内藤記念科学奨励金 研究助成
2021 ブレインサイエンス振興財団 研究助成
2021 光科学技術振興財団 研究助成
2021 せりか基金 研究助成金
2021 上原記念生命科学財団 研究助成金
2020 「小出良夫基金」研究奨励金
2020 国立遺伝学研究所共同研究「NIG-JOINT」共同研究(B)
2018 加藤記念難病研究助成基金 研究助成
2018 せりか基金 研究助成金
2016 日本科学協会: 海外発表促進助成金
2014 「生命の彩」ALS研究助成基金助成金
2013 武田科学振興財団 医学系研究奨励
2013 ノバルティス科学振興財団(ノバルティス研究奨励金)
2012 第一三共生命科学研究振興財団(研究助成)
2012 住友財団(基礎科学研究助成)
2011 総研大学融合推進センター(若手研究者研究支援事業)
2011 三菱財団(自然科学研究助成)
2010 総研大学融合推進センター(若手研究者研究支援事業)
2010 花王芸術・科学財団(科学技術研究助成)
2009 上原記念生命科学財団(研究奨励金)
2005 日本科学協会(笹川科学研究助成)
